Necroptosis
When cells become largely dysfunctional, they self-destruct. This process in neurons (brain cells) is called ‘necroptosis’. The recent discovery, made by researchers at the UK’s Dementia Research Institute at University College London[1], showed that an abnormal accumulation of a proteinProteins are large molecules consisting of chains of amino acids. Proteins are essential nutrients for the human body – they are a building block of… called tau, that then becomes phosphorylated, making it tangled and dysfunctional, triggers a specific molecule called MEG3 that triggers brain cell death.
“For the first time we get a clue as to how and why neurons die in Alzheimer’s disease. There’s been a lot of speculation for 30-40 years, but nobody has been able to pinpoint the mechanisms. It really provides strong evidence it’s this specific suicide pathway.” researcher Prof Bart De Strooper, from the UK’s Dementia Research Institute told the BBC.
P-tau
Even before this too much phosphorylated-tau, abbreviated to p-tau, messes up the cell’s energy factories called mitochondria, potentially leading to brain fatigue. The more p-tau that accumulates, the greater becomes a person’s risk for cognitive problems and Alzheimer’s dementia. Also, those with memory decline have been shown to have relatively more p-tau to tau protein.
The critical prevention question is what stops too much of the tau protein from turning into the potentially harmful p-tau, and what helps restore p-tau to normal tau protein.
B Vitamins and Homocysteine
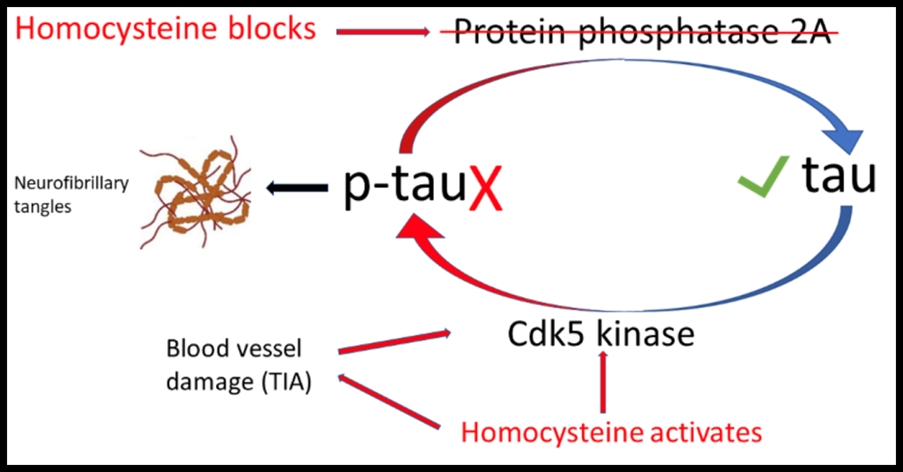
The answer is remarkably simple – a lack of B vitamins and raised blood levels of homocysteineHomocysteine is an amino acid found in the blood. Elevated levels of homocysteine have been associated with narrowing and hardening of the arteries, an increased… – an established risk factor for memory decline, measurable in the blood.
When levels of B vitamins (B6, B12 and folate) are low, blood levels of homocysteine go up. This activates one enzyme (Cdk5 kinase) that adds the bad ‘p’ to tau and blocks another enzyme (protein phosphatase A2) which removes the dangerous ‘p’ restoring normal tau protein.[2] [3]
High homocysteine also damages the tiny blood vessels in the brain, leading to ‘mini-strokes’ or transient ischemic attacks (TIAs), which further raises levels of p-tau. Homocysteine both raises levels of the dangerous p-tau[4] and can also bind to tau [5], further generating the neurofibrillary tangles that then trigger brain cell death.
So, the simplest way to stop the formation of p-tau, and neurofibrillary tangles, and keep your brain healthy, is to keep your plasma homocysteine level below 10mcmol/l. Half of those above 65 have a level of homocysteine higher than this.
Supplementing B Vitamins
The easiest way to do that is to supplement B6, B12 and folate. But it’s good to eat greens and beans, high in folate. B12 is only in animal foods – meat, seafood, eggs and milk.
While an optimal supplemental intake for a middle-aged person might be 20mg of B6, 10mcg of B12, and 400mcg of folate, many older people start to dramatically lose their ability to absorb B12, the absorption of which requires stomach secretions.
Antacid ‘PPI’ medication such as omeprazole accelerates this decline, promoting B12 deficiency. After over four years of use, dementia risk increases by a third.[6] Then, as studies show, you might need a lot more B12, such as 500mcg, to get a little more into your bloodstream, and possibly more supplemental folate, in the region of 500 to 800mcg.
This is the cheapest, safest and most logical solution to lowering p-tau and preventing brain cells committing suicide. The problem is that these nutrients, invented by nature, cannot be patented. Therefore, it is not in the interest of the pharmaceutical industry to research them.
Consequently, drugs are being developed and tested that block the kinase enzyme and activate the phosphatase enzyme[7], which is exactly what the homocysteine-lowering B vitamins do.
But, so far, there are no human clinical drug trials reporting significant benefit. On the other hand, trials giving these kind of doses of B6, B12 and folic acidWhat it does: Critical during pregnancy for the development of a baby’s brain and nerves. Also essential for brain and nerve function. Needed for utilising…, have shown up to two thirds slower rate of brain shrinkage[8] and virtually no further memory loss in those with pre-dementia[9].
References
[1] https://www.science.org/doi/10.1126/science.abp9556 Balasu S et al. Science
14 Sep 2023 Vol 381, Issue 6663 pp. 1176-1182 DOI: 10.1126/science.abp9556
[2] Smith AD, Refsum H. Homocysteine, B Vitamins, and Cognitive Impairment. Annu Rev Nutr. 2016 Jul 17;36:211-39. doi: 10.1146/annurev-nutr-071715-050947. PMID: 27431367.
[3] LiJ-G,ChuJ,BarreroC,MeraliS,Pratico`D.2014.Homocysteine exacerbatesβ-amyloid, tau pathology, and cognitive deficit in a mouse model of Alzheimer’s disease with plaques and tangles. Ann. Neurol. 75:851–63
[4] Shirafuji N et al Homocysteine Increases Tau Phosphorylation, Truncation and Oligomerization. Int J Mol Sci. 2018 Mar 17;19(3):891. doi: 10.3390/ijms19030891. PMID: 29562600; PMCID: PMC5877752.
[5] Bossenmeyer-Pourié C et al. N-homocysteinylation of tau and MAP1 is increased in autopsy specimens of Alzheimer’s disease and vascular dementia. J Pathol. 2019 Jul;248(3):291-303. doi: 10.1002/path.5254. Epub 2019 Mar 19. PMID: 307349
[6] Northuis C, Bell E, Lutsey P, George KM, Gottesman RF, Mosley TH, Whitsel EA, Lakshminarayan K. Cumulative Use of Proton Pump Inhibitors and Risk of Dementia: The Atherosclerosis Risk in Communities Study. Neurology. 2023 Aug 9:10.1212/WNL.0000000000207747. doi: 10.1212/WNL.0000000000207747. Epub ahead of print. PMID: 37558503.
[7] Xia, Y., Prokop, S. & Giasson, B.I. “Don’t Phos Over Tau”: recent developments in clinical biomarkers and therapies targeting tau phosphorylation in Alzheimer’s disease and other tauopathies. Mol Neurodegeneration 16, 37 (2021). https://doi.org/10.1186/s13024-021-00460-5
[8] Smith AD, Refsum H. Homocysteine, B Vitamins, and Cognitive Impairment. Annu Rev Nutr. 2016 Jul 17;36:211-39. doi: 10.1146/annurev-nutr-071715-050947. PMID: 27431367; see also Jernerén F., et al. Am J Clin Nutr. 2015 Jul;102(1):215-21.

Comments
Join the Conversation on our Facebook Page