One in a 100 cases ‘in the genes’
Just one in 100 cases of Alzheimer’s are caused by mutations in genes (presenilin, APP) which account for a most of the familial, early onset cases. [iv] There are genes, notably the ApoE 4 gene variant, which can increase risk but the odds can be mitigated by changes in diet and lifestyle[v] One often hears that having the ApoE4 gene allele increases risk by 30%, but that is the relative risk increase, not the absolute risk increase, which is 4%[vi] – perhaps less if you do all the right things.
Changes in the brain start happening up to 30 years before a diagnosis of mild cognitive impairment (MCI), dementia or Alzheimer’s. There is no way to reverse the brain mass loss found in those with Alzheimer’s. Therefore, early diagnosis of subtle cognitive decline to identify those at risk is vital, followed by preventive action to reduce that risk.
Prevention is the solution – not genes nor drugs
There are diseases, such as diabetes, hypertensionHypertension is more commonly known as high blood pressure…. and stroke, which increase risk but our experts have the view that these diseases primarily are not the cause but the consequence of diet and lifestyle factors.
Lowering high blood pressure is an example. A recent study[vii] in almost ten thousand over 50 year olds with high blood pressure, using either standard hypertensive medication to reduce high ‘systolic’ blood pressure to the usual level of below 140 versus intensive medication to get blood pressure below 120 found a modest, non-significant difference in the number who developed dementia (149 vs 176). This study was not significant yet reported a non-significant 20% ‘relative’ reduced risk with intensive medication versus standard treatment. This is rather misleading. In addition, there was no placebo group so we don’t even know if this apparent reduction was better or worse than doing nothing. Nor did they mention adverse effects. These were reported in an earlier trial if you went looking. In terms of absolute risk reduction, if one thousand people were treated, that would be the number needed to potentially save one person from an MCI diagnosis – 380 would have a potentially life-threatening ‘serious adverse effect’ meaning they would either die or require prolonged hospitalisation.
The best meta-analysis of 396 trials, by Professor Jin Tai Yu from the University of Fudan, Shanghai, who is a member of our Scientific Advisory Board, confirm the potential impact of risk reduction. The number one most effective modifable risk factor was homocysteine-lowering B vitamins.[viii] The National Institutes of Health meta-analysis attributed 22% of the risk to homocysteineHomocysteine is an amino acid found in the blood. Elevated levels of homocysteine have been associated with narrowing and hardening of the arteries, an increased… and 22% of the risk to low seafood/omega-3 intake.[ix]
Professor David Smith’s group at Oxford University (another member of our SAB) found 73% less brain shrinkage, no further memory decline and 30% of the pre-dementia (MCI) trial participants ending the one year trial with a clinical dementia rating of zero, if they had sufficient omega-3 blood level and took the homocysteine lowering B vitamins, versus placebo.[x] To put this into context the best drug to date has reported 2% less brain shrinkage and no change in clinical dementia rating. See image, used with permission from Douaud (yellow denotes area of significant atrophy)
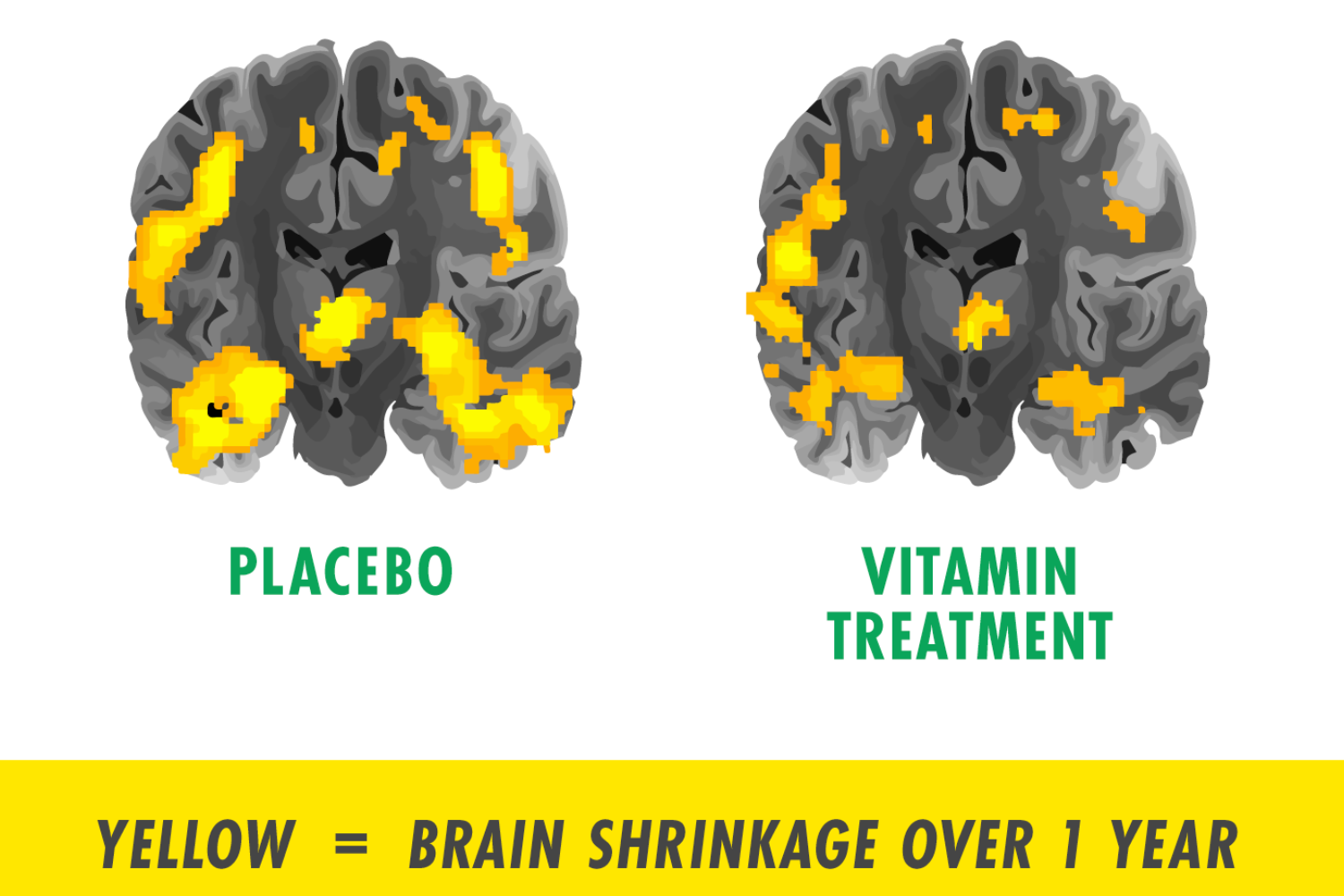
Ref: Jernerén F, Elshorbagy AK, Oulhaj A, Smith SM, Refsum H, Smith AD. Brain atrophy in cognitively impaired elderly: the importance of long-chain ω-3 fatty acids and B vitamin status in a randomized controlled trial. Am J Clin Nutr. 2015 Jul;102(1):215-21. doi: 10.3945/ajcn.114.103283. Epub 2015 Apr 15. PMID: 25877495 – https://pubmed.ncbi.nlm.nih.gov/25877495/
Take the free Cognitive Function test online
In 2011, with the help of Dr Celeste De Jager Loots, an expert in neuropsychological assessment (also in our SAB), we digitised and validated the standard Cognitive Function Test used to assess cognitive health and screen for MCI and dementia and made it available, for free, online.[xi] To date over 370,000 have taken the test demonstrating huge appetite for this sort of tool. The Cognitive Function Test measures the critical aspects that indicate risk: episodic memory, attention and executive function. Decline in these aspects of cognition, plus an accelerated rate of brain shrinkage, are the hallmarks of cognitive impairment.
In 2018 we introduced a risk factor questionnaire and commenced a research project to inform the building of an interactive, effective means of changing key diet and lifestyle behaviours to reduce risk and asked University College London and NHS researchers to test it, and explore barriers and weaknesses to increase compliance and behaviour change. They reported that 88% found in useful and 37% made diet or lifestyle changes as a result. Our goal is to substantially improve these percentages.[xii] This year, working with our excellent team of scientific advisors whom you can meet here: foodforthebrain.org/SAB, including Professor Robert Lustig – our ‘sugar’ man – we launched the Dementia Risk Index (DRI). The questionnaire only covers things you can change and works out your risk across eight domains. If you score 100% you are doing everything ‘wrong’, and if you score 0% you are doing everything right to reduce your risk.
Upgrade your brain with COG-NITION
Together, we need to educate, break down barriers and get people to join our grassroots campaign by taking the Cognitive Function Test at www.foodforthebrain.org, filling in the Dementia Risk Index and joining our citizen’s health community, to really show that Alzheimer’s is Preventable.
Revolutions also happen bottom up, not top down. Please share the Alzheimer’s is Preventable Manifesto at www.foodforthebrain.org/AIPmanifesto.
The evidence for the 8 domains of Alzheimer’s prevention
Read the evidence for the 8 domains of Alzheimer’s prevention here.
References
[i] Ackley SF, Zimmerman SC, Brenowitz WD, Tchetgen Tchetgen EJ, Gold AL, Manly JJ, et al. Effect of reductions in amyloid levels on cognitive change in randomized trials: instrumental variable meta-analysis. BMJ. 2021; 372: n156.
[ii] Editorial. Dementia: a false promise. Lancet 2014;384(9948):1072
[iii] Cummings JL, Goldman DP, Simmons-Stern NR, Ponton E. The costs of developing treatments for Alzheimer’s disease: A retrospective exploration. Alzheimer’s Dement. 2022;18:469–477. https://doi.org/10.1002/alz.12450
[iv] Bekris L et al, ‘Genetics of Alzheimer disease’ Journal of Geriatric Psychiatry and Neurology 2010, 23(4) 213-227
[v] Solomon A, Turunen H, Ngandu T, Peltonen M, Levalahti E, Helisalmi S, et al. Effect of the Apolipoprotein E Genotype on Cognitive Change During a Multidomain Lifestyle Intervention: A Subgroup Analysis of a Randomized Clinical Trial. JAMA Neurol. 2018; 75: 462-70; also see ref 5.
[vi] SPRINT MIND Investigators for the SPRINT Research Group, Williamson JD, Pajewski NM, Auchus AP, Bryan RN, Chelune G, Cheung AK, Cleveland ML, Coker LH, Crowe MG, Cushman WC, Cutler JA, Davatzikos C, Desiderio L, Erus G, Fine LJ, Gaussoin SA, Harris D, Hsieh MK, Johnson KC, Kimmel PL, Tamura MK, Launer LJ, Lerner AJ, Lewis CE, Martindale-Adams J, Moy CS, Nasrallah IM, Nichols LO, Oparil S, Ogrocki PK, Rahman M, Rapp SR, Reboussin DM, Rocco MV, Sachs BC, Sink KM, Still CH, Supiano MA, Snyder JK, Wadley VG, Walker J, Weiner DE, Whelton PK, Wilson VM, Woolard N, Wright JT Jr, Wright CB. Effect of Intensive vs Standard Blood Pressure Control on Probable Dementia: A Randomized Clinical Trial. JAMA. 2019 Feb 12;321(6):553-561. doi: 10.1001/jama.2018.21442. PMID: 30688979; PMCID: PMC6439590.
[vii] https://pubmed.ncbi.nlm.nih.gov/30688979/
[viii] Yu JT, Xu W, Tan CC, Andrieu S, Suckling J, Evangelou E, Pan A, Zhang C, Jia J, Feng L, Kua EH, Wang YJ, Wang HF, Tan MS, Li JQ, Hou XH, Wan Y, Tan L, Mok V, Tan L, Dong Q, Touchon J, Gauthier S, Aisen PS, Vellas B. Evidence-based prevention of Alzheimer’s disease: systematic review and meta-analysis of 243 observational prospective studies and 153 randomised controlled trials. J Neurol Neurosurg Psychiatry. 2020 Nov;91(11):1201-1209. doi: 10.1136/jnnp-2019-321913. Epub 2020 Jul 20. PMID: 32690803; PMCID: PMC7569385.
[ix] Beydoun M et al, ‘Epidemiologic studies of modifiable factors associated with cognition and dementia: systematic review and meta-analysis’ BMC Public Health 2014, 14:643
[x] Smith AD, Refsum H. Homocysteine, B vitamins, and cognitive impairment. Annu Rev Nutr. 2016; 36: 211-39
[xi] Trustram Eve C, de Jager CA. Piloting and validation of a novel self-administered online cognitive screening tool in normal older persons: the Cognitive Function Test. Int J Geriatr Psychiatry. 2014 Feb;29(2):198-206. doi: 10.1002/gps.3993. Epub 2013 Jun 11. PMID: 23754255.
[xii] Bird et al., 2021. The psychological determinants of making lifestyle and dietary behaviours after using an online cognitive health tool and its associated recommendations for protective cognitive health behaviours. Eur Journ Psych, 35 (3), 145-156. https://doi.org/10.1016/j.ejpsy.2021.02.001

Comments
Join the Conversation on our Facebook Page